Why Biocelk?
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Explore Treatments
Scientific Literature
Featured Treatments
Wellness & Longevity
Dermo-aesthetic

Fertility
About Biocelk
Our Brands
Visit: regenivf.com
Therapies for Multiple Conditions
The human body has a natural capacity for repair and balance. However, over time, chronic diseases, inflammatory processes, or injuries can affect this capacity, leading to functional deterioration and a decline in quality of life.
At Biocelk®, we address these conditions through regenerative medicine, using protocols designed to act on different systems of the body by supporting key biological processes such as tissue regeneration, modulation of inflammation, and cellular communication.
This approach makes it possible to treat multiple conditions in an integrated way, focusing not only on the symptom, but also on the biological environment from which it originates.
Clinical Benefits
At Biocelk®, we bring together medical innovation and regenerative medicine to offer you a more comprehensive alternative for a wide range of conditions and health needs.
Degenerative Diseases
Improvement in pain, stiffness, and function in osteoarthritis
Reduced pain and improvement in patients with knee osteoarthritis
When every step hurts and stiffness limits your daily mobility, finding an effective alternative becomes a priority. Amado Pico and collaborators published a randomized, double-blind, controlled pilot study in Cytotherapy (2025), where they evaluated a single intra-articular application of a product based on umbilical cord mesenchymal stem cells versus a corticosteroid in patients with knee osteoarthritis. The results showed significant improvement in pain, stiffness, and function (WOMAC), as well as in pain scale and quality of life (SF-36), over 12 months in the cellular treatment group, with no serious adverse events.
In practical terms, this translates into greater freedom of movement, less pain when walking, and a better quality of life in everyday activities, even with a single application.
Alzheimer’s Disease
Measurable reduction in biomarkers associated with Alzheimer’s disease after treatment
When a neurodegenerative disease progresses, one of the greatest concerns is whether anything can influence the biological processes that sustain it, beyond symptomatic management. Lee and collaborators published a phase IIa trial in Dementia and Neurocognitive Disorders (2025) in patients with Alzheimer’s disease, where they evaluated intraventricular administration of a mesenchymal cellular component and, in an open-label extension, its combination with dexamethasone. Although no significant clinical improvements versus placebo were observed, reductions were documented in cerebrospinal fluid biomarkers related to Alzheimer’s, such as amyloid beta, total tau, and phosphorylated tau. In addition, dexamethasone appeared to reduce immune reactions such as fever, vomiting, and increases in white blood cells and interleukin-6.
This suggests an interesting biological signal regarding the disease, while also highlighting the need to better control the inflammatory response to treatment.
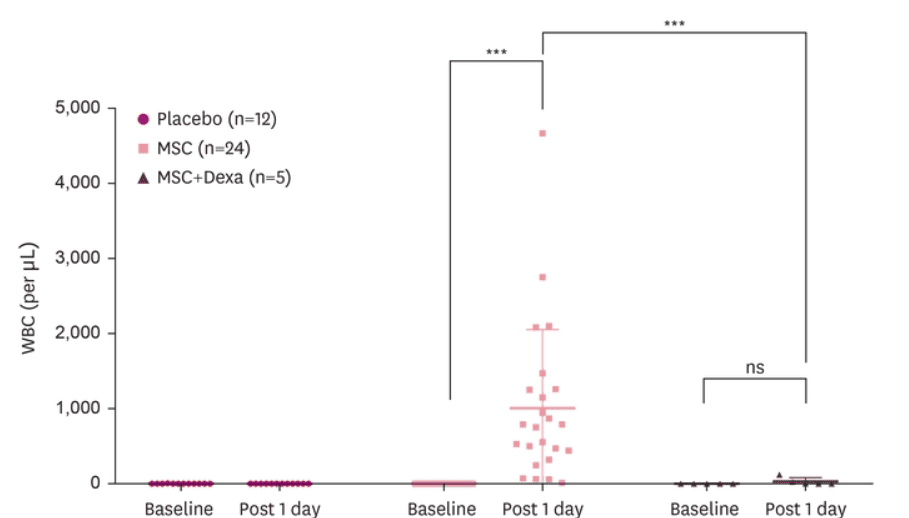
The white blood cell count in the cerebrospinal fluid rises markedly after cellular administration, showing an inflammatory response. However, in the dexamethasone group, this increase is notably lower, visually confirming a modulating effect on the immune response.
Parkinson’s Disease
- Functional improvements observed without adverse effects after repeated stem cell infusions
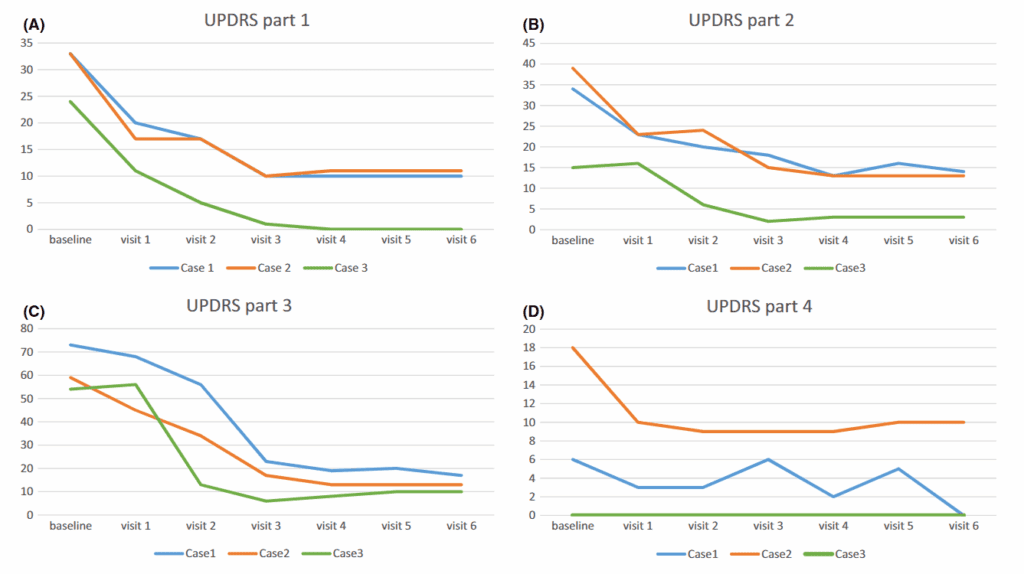
Figure 1A, which evaluates non-motor aspects such as mental state and behavior, shows an overall tendency toward lower scores in all three cases, suggesting improvement in these domains over time.
Figure 1B, related to daily living activities, also shows a progressive reduction in scores at several follow-up points, indicating possible improvement in functional independence.
Figure 1C, which evaluates motor function, shows one of the most relevant changes, with lower scores in all three patients, suggesting improvement in motor symptoms such as rigidity, tremor, or slowness.
Figure 1D, which evaluates treatment complications, shows scores that remain low or trend downward, indicating stability or reduction of complications.
As Parkinson’s disease progresses, rigidity, slowness of movement, and dependence on medication often deeply affect quality of life. Shigematsu and collaborators, in Acta Neurologica Scandinavica (2022), evaluated repeated administration of adipose tissue-derived mesenchymal stem cells in patients with this disease through a monthly intravenous infusion schedule. During follow-up, no adverse effects were reported and all patients showed improvement on the MDS-UPDRS scale, used to measure the severity of Parkinson’s disease; in addition, two patients reported feeling “much better,” and one was able to significantly reduce medication.
This cellular approach may positively influence neurological function and the patient’s day-to-day experience, without compromising short-term safety.
Schedule a medical evaluation and discover how Biocelk® can design a strategy tailored to the complexity of your case.
Lupus
83% of patients achieved clinical improvement after cellular therapy with a favorable safety profile
When systemic lupus erythematosus does not respond to conventional treatments, uncertainty and persistent symptoms can deeply affect quality of life. Kamen and collaborators published a phase I trial in Lupus Science & Medicine (2022) in refractory patients, where they evaluated intravenous administration of a cellular component derived from umbilical cord tissue. After a single infusion, five out of six patients met the clinical response criterion (SRI-4), with minimal adverse events. At the biological level, reductions were observed in B-cell subpopulations associated with lupus activity, together with an increase in GARP-TGFβ complexes correlated with clinical improvement.
This type of therapy may measurably modulate the immune system, aligning with improvement in disease activity in complex patients.
Multiple Sclerosis
Years of stability and functional improvement in a case of progressive multiple sclerosis
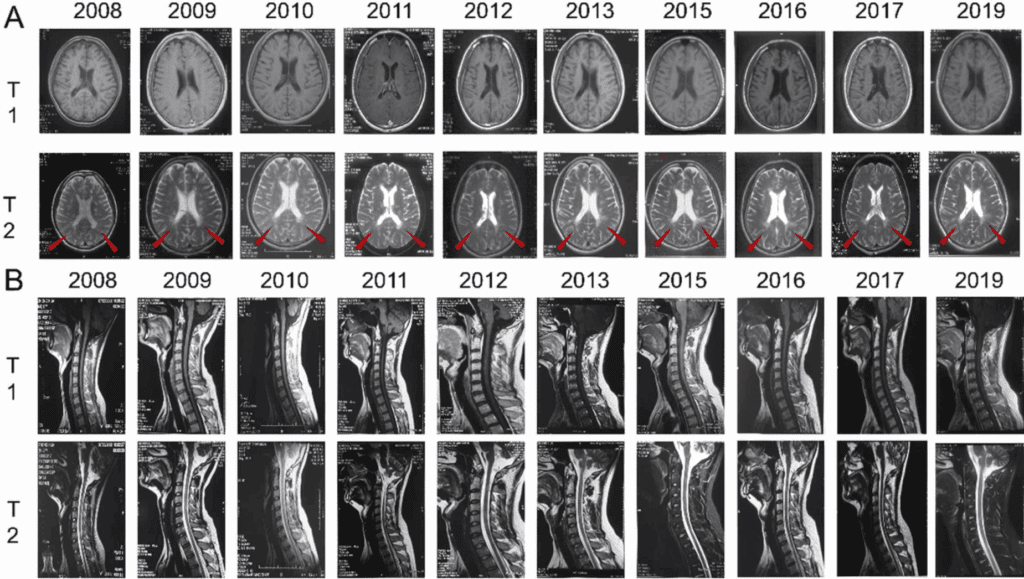
As multiple sclerosis progresses, loss of mobility, fatigue, and constant relapses can deeply limit daily life. Liu and collaborators reported in Clinical Neurology the follow-up of a patient with progressive multiple sclerosis (2022) treated with multiple infusions of a mesenchymal cellular component over more than a decade.
Over 10 years, the EDSS score decreased steadily until reaching 1.0, while MRI imaging showed no new lesions; in addition, the treatment was well tolerated with no relevant adverse effects. After therapy was discontinued, the EDSS score increased again and new lesions appeared.
Cellular therapy may be associated with prolonged disease stabilization while treatment is maintained, reflected in less clinical and radiologic progression.
The figure shows a longitudinal comparison of MRI studies between 2008 and 2019, allowing visualization of the structural evolution of the disease over time. In the initial images, multiple demyelinating plaques can be seen in periventricular, subcortical, and corpus callosum regions, characteristic of active multiple sclerosis. As the years pass under treatment, the images reflect a decrease in the number and size of the lesions, as well as reduced edema around them, indicating lower inflammatory activity. In addition, some lesions take on an inactive appearance, suggesting stability in the pathological process. However, after treatment suspension, the later images show the reappearance of new lesions, which coincides with the clinical deterioration observed.
Heart Failure
Improvement in cardiac function with stem cells in patients with chronic heart failure
Living with heart failure often feels like a constant limitation: fatigue, shortness of breath on exertion, and concern that, even with optimal treatment, the heart may not recover its strength. Bartolucci and collaborators published the RIMECARD trial in Circulation Research (2017), a randomized study that evaluated intravenous infusion of umbilical cord-derived mesenchymal stem cells in patients with stable chronic heart failure and reduced ejection fraction. Participants received a dose of 1×10^6 cells/kg or placebo, and the results showed that only the treated group had significant improvement in ejection fraction at 3, 6, and 12 months (+7.07% vs +1.85% in placebo).
In this group of patients, stem cell therapy contributed to measurable improvement in cardiac function, functional class, and quality of life, with no serious complications or treatment-related adverse effects. If you live with heart failure and want to explore new treatment alternatives, schedule a consultation to evaluate whether this approach may be appropriate for you.
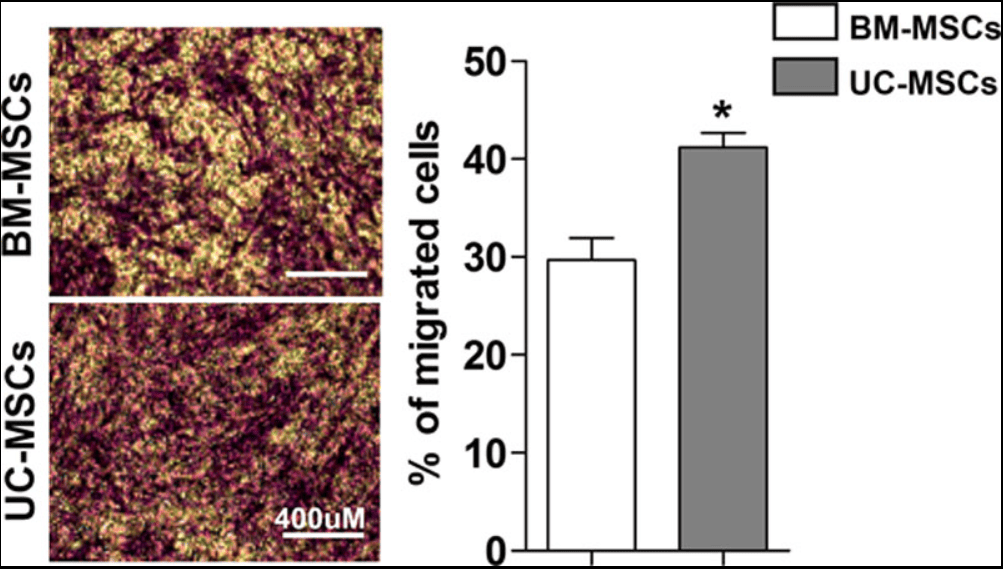
Transwell migration assay comparing UC-MSCs vs BM-MSCs in response to serum from patients with HFrEF (16 h). Includes a representative staining image (crystal violet) and quantification of the percentage of migrated cells, showing greater migration for UC-MSCs in this experimental context.
When your health needs to be taken care of from a broader scope, Biocelk® offers a different way to join you.
Cirrhosis
A new opportunity to improve liver function and prolong life in advanced cirrhosis
The liver may recover part of its functional capacity, and the patient may experience a more stable and prolonged course over time.
If you are looking for a regenerative medicine-based alternative today, at Biocelk we develop advanced therapies designed to support liver recovery; you can take the next step and explore whether this is the right approach for your case.
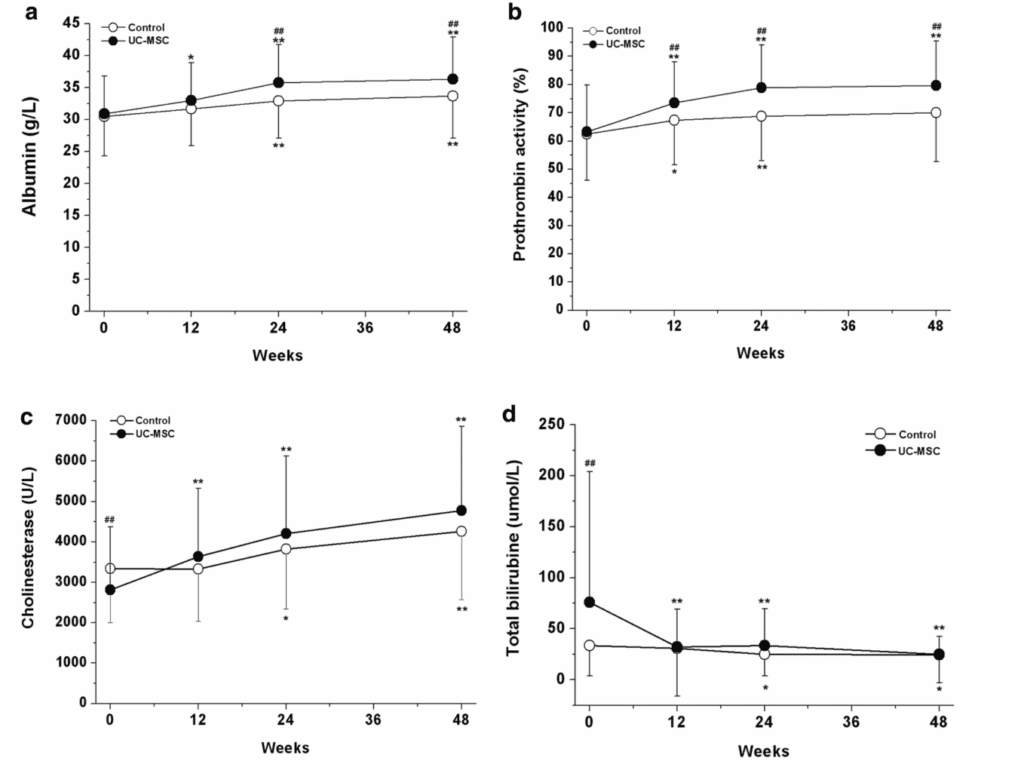
The figure evaluates liver function and shows progressively favorable changes: parameters related to the liver’s ability to synthesize proteins and coagulate increase (such as albumin and prothrombin activity), while markers of liver injury such as bilirubin decrease. Visually, these graphs reflect a clear trend toward stabilization and improvement in liver status.
Autoimmune and Infectious Diseases
Rheumatoid Arthritis
Sustained improvement in pain, inflammation, and joint function is now possible in patients with rheumatoid arthritis thanks to Biocelk
Living with constant pain, morning stiffness, and limited movement can deeply affect your quality of life. Wang and collaborators, from the Cell Therapy Center of the Air Force Hospital of the People’s Liberation Army (2019), evaluated the effect of umbilical cord-derived cellular therapies in patients with rheumatoid arthritis, combined with conventional treatment. Over 1 to 3 years of follow-up, they observed a significant reduction in inflammatory markers such as CRP, ESR, and rheumatoid factor, as well as improvement in disease activity and physical function, reflected in lower DAS28 and HAQ scores.
Inflammation may be better controlled, pain may decrease, and regaining mobility becomes a real possibility in daily life.
If you are looking for an alternative that goes beyond symptom control, at Biocelk we develop advanced therapies focused on modulating inflammation and supporting joint recovery.
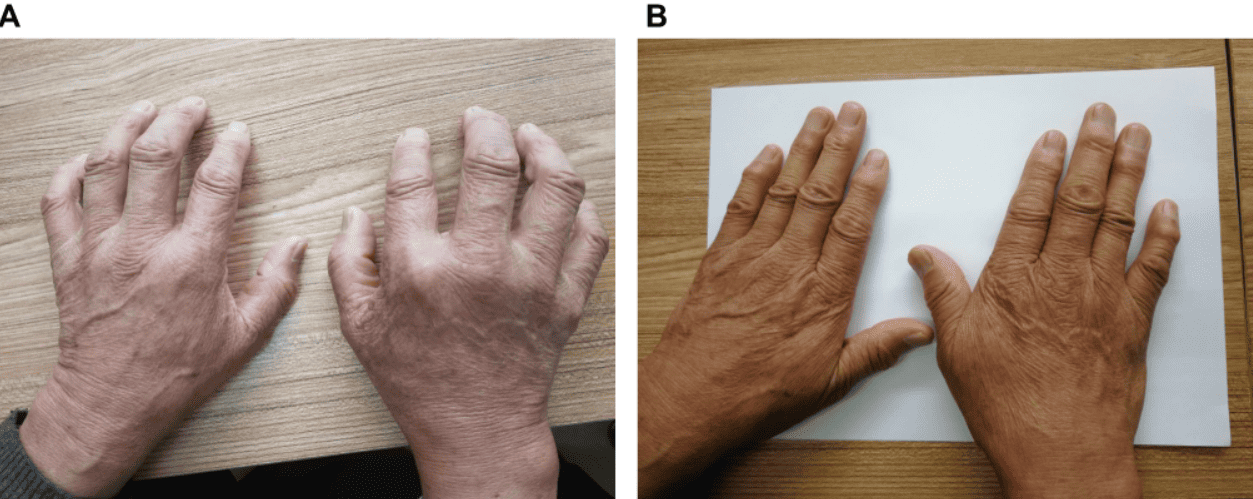
The figure shows a patient with clear hand deformity, unable to fully extend the fingers due to progressive joint damage. After treatment, the image shows notable recovery in hand alignment and mobility, with disappearance of pain and functional improvement that allows the patient to resume everyday physical activities.
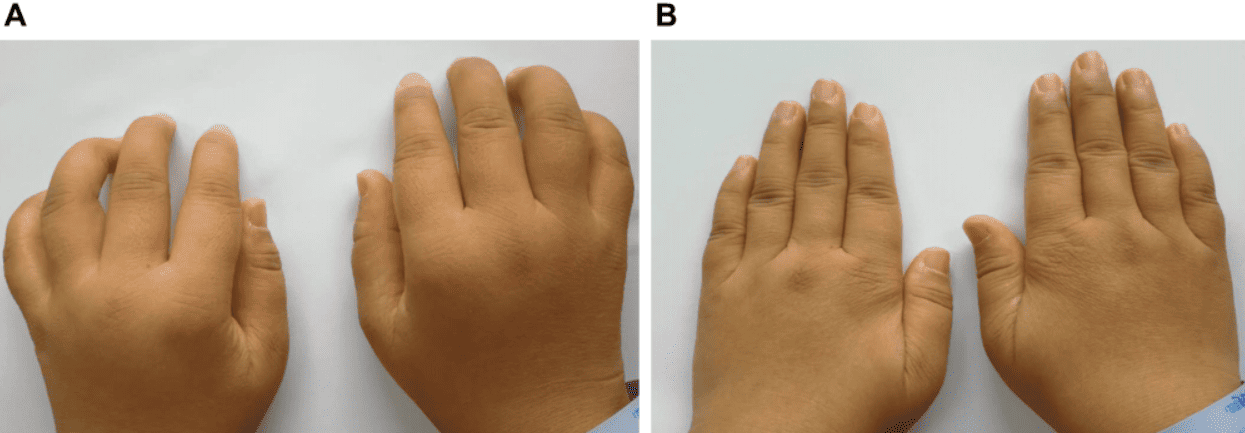
The figure shows a patient with severe inflammation, stiffness, and difficulty closing the hands. After intervention, visible reduction in inflammation and improved mobility are evident, allowing movements that were previously limited.
Crohn’s Disease
Biocelk: A cellular alternative with encouraging results in Crohn’s disease
These findings show relevant clinical improvement and additional support in disease control with mild side effects. The treatment helped patients feel better and require fewer steroids.
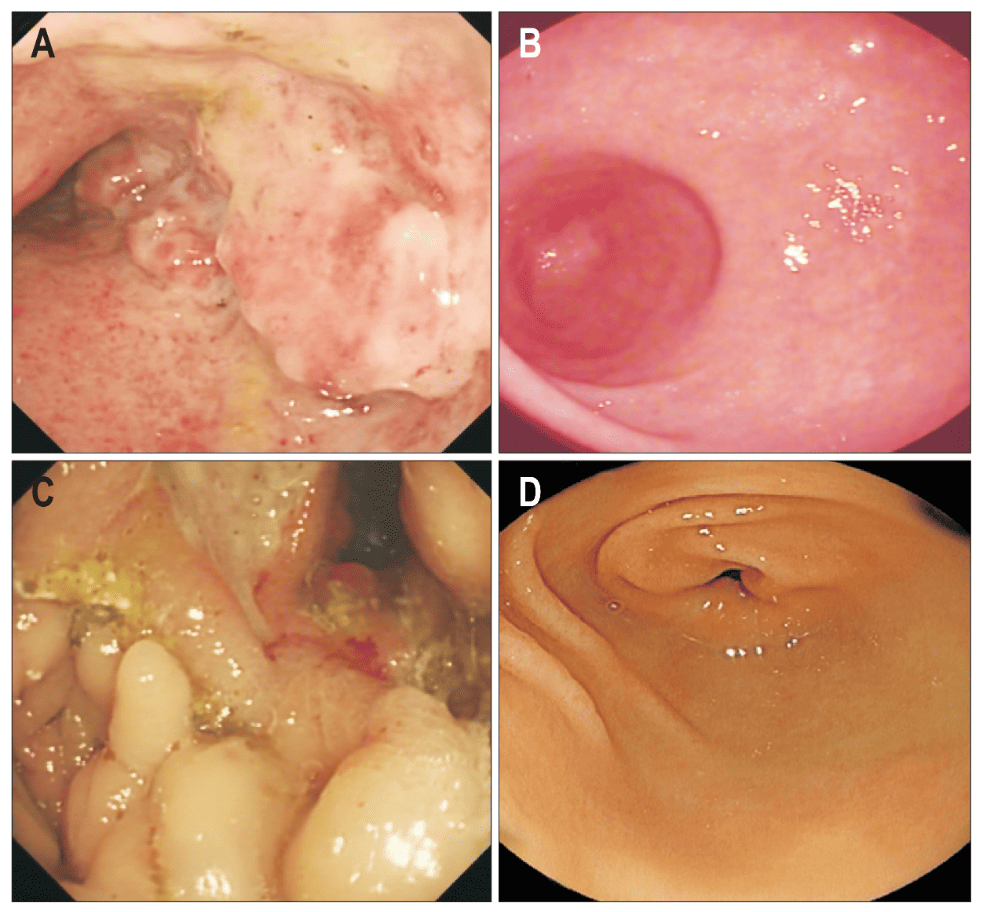
The figure shows a particularly valuable finding: at 12 months after treatment, colonoscopy revealed remarkable mucosal recovery compared with the initial images. In the article by Zhang and team, published in Gut and Liver in 2018, the panels corresponding to two patients allow direct observation of visual improvement in intestinal tissue after the use of umbilical cord mesenchymal stem cells. Beyond the clinical indices, this image adds something very powerful: it shows that the benefit was reflected not only in numbers, but also in the appearance of the mucosa evaluated by colonoscopy. This is favorable because it suggests a reduction in visible inflammatory damage and a better condition of intestinal tissue throughout follow-up.
Ulcerative Colitis
- Clinical response and remission with a favorable safety profile in ulcerative colitis
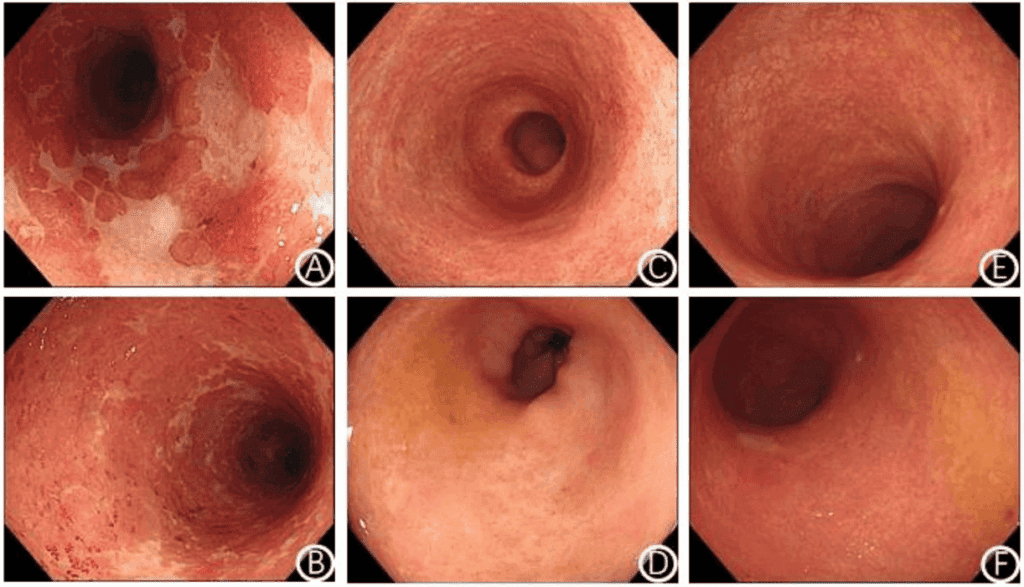
The figure shows favorable progression of intestinal tissue after treatment with umbilical cord mesenchymal stem cells. In the endoscopic images, the initial condition gives way to a progressively more stable appearance at 2 and 6 months, accompanied by reduced colonic inflammation and disappearance of ulcers, as reported in the study. In addition, the authors describe gradual progress toward mucosal remission and less inflammatory infiltration in histopathologic findings. This is relevant because it suggests that the improvement was not only perceived in symptoms, but also in the appearance of intestinal tissue directly evaluated.
These results suggest a favorable effect on intestinal inflammation and the patient’s general condition, with improvement reflected not only in clinical studies, but also in the way the body regains balance. In simple terms: there were patients who were better, with objective signs of improvement. At Biocelk, you can explore whether this cellular approach may be an alternative worth considering in your case.
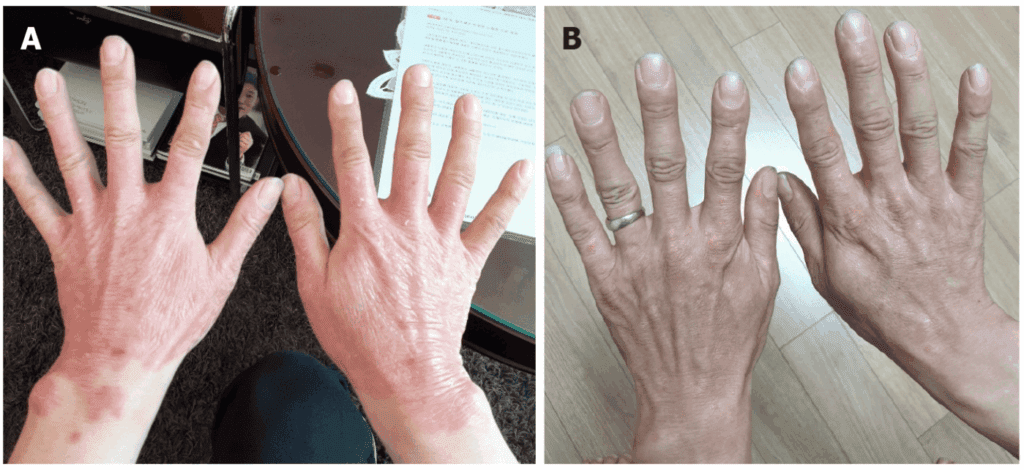
The figure shows favorable and easily appreciable progression of psoriatic lesions after treatment with mesenchymal stem cells derived from umbilical cord tissue. Throughout follow-up, the redness progressively decreased until it had almost disappeared by day 122 after the first intervention, with visible improvement in the hands, wrists, and ear. This finding is especially valuable because it allows direct observation of skin with less visible inflammation and a much more stable appearance than at baseline.
Psoriasis
Visible improvement in psoriasis and quality of life
This showed a significant reduction in visible psoriasis activity together with marked improvement in how the patient felt and experienced daily life.
Pneumonia and Inflammatory Conditions
Rapid improvements in lung function with stem cells in severe COVID-19 pneumonia cases
These results point to a positive effect of stem cells on immune modulation and inflammation reduction, which may contribute to faster recovery in severe pneumonia cases. If you are considering alternatives as part of your treatment, a medical consultation can help determine whether this approach may be appropriate for you.
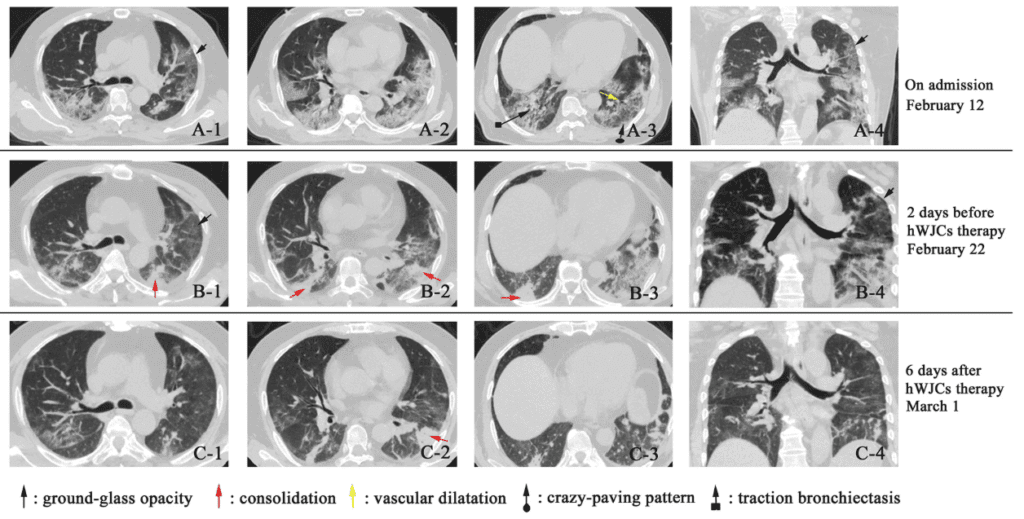
Series of CT scans comparing the patient’s pulmonary progression with COVID-19.
A-1 to A-4 (Feb 12): ground-glass opacities and bilateral pneumonic infiltration; regions with traction bronchiectasis are indicated; in the left lower lobe, a crazy-paving pattern is described (ground-glass opacity with septal thickening), with scattered areas of consolidation and vascular dilation.
B-1 to B-4 (Feb 22): slight symptomatic improvement, but significant pneumonia remains; the initial ground-glass regions decrease and new subpleural consolidation appears.
C-1 to C-4 (Mar 1, 6 days post-infusion): pneumonic infiltration is markedly reduced; most opacities clear or disappear; partial areas of consolidation persist.
Type 1 Diabetes
A new possibility to improve Type 1 diabetes control
Better preserving beta-cell function during the first year after diagnosis may make an important difference for people living with Type 1 diabetes who depend on insulin from very early stages. Lu and team published a controlled clinical study in Stem Cell Research & Therapy (2021) in which they evaluated double intravenous administration of allogeneic mesenchymal stromal cells derived from umbilical cord tissue in people with recent-onset Type 1 diabetes. After one year of follow-up, 40.7% of treated patients achieved clinical remission, a significantly higher proportion than in the control group, and three adults achieved insulin independence for periods ranging from 3 to 12 months; in addition, no serious adverse effects were observed.
These results suggest better preservation of endogenous insulin production and a favorable biological response at a key stage of the disease. In simple terms, some patients preserved pancreatic function better, and some even went for a period without needing insulin.
One of the most encouraging findings of the study is shown here: compared with baseline and one-year follow-up, more patients treated with umbilical cord mesenchymal cells preserved or improved their C-peptide levels compared with the control group. In adults in particular, postprandial response showed a more favorable course in the treated group. This is relevant because C-peptide is a sign that the body still retains the ability to produce its own insulin.
Type 2 Diabetes
More time in range and better glucose control with cellular therapy
When glucose levels rise and fall constantly, managing Type 2 diabetes becomes more difficult, and concerns about the short- and long-term impact on health increase. Zang and team published a phase II randomized, placebo-controlled trial in Stem Cells Translational Medicine (2023), in which they evaluated umbilical cord-derived mesenchymal stem cells in adults with Type 2 diabetes. After three intravenous infusions and 48 weeks of follow-up, the treated group showed a more marked improvement than placebo in both time in range and HbA1c, with changes of 21.36 vs. 6.32 in TIR and −1.36 vs. −0.51 in HbA1c; in addition, more patients reached the goal of TIR ≥ 70% and HbA1c < 7% at weeks 9 and 48.
These findings suggest more stable glycemic control and a more favorable reduction in glucose variability, something especially valuable for living with greater metabolic balance. Some patients were able to keep their glucose better controlled for longer.
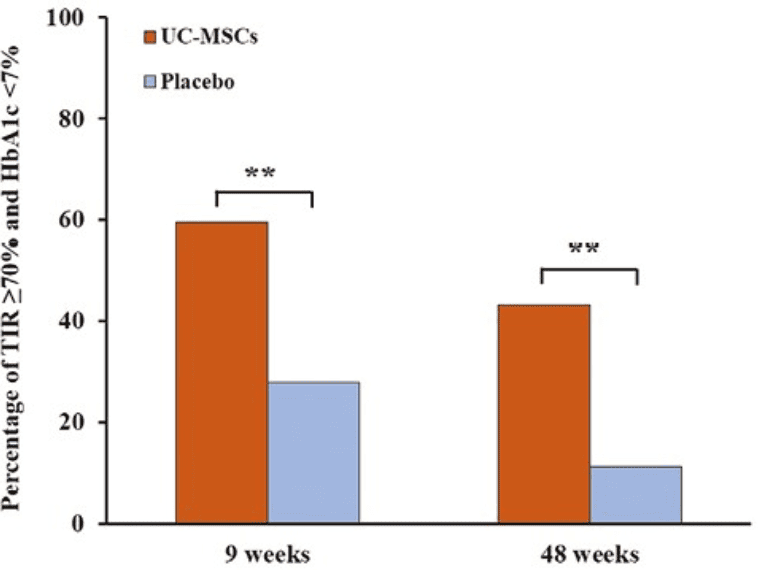
The figure shows that the group treated with umbilical cord-derived mesenchymal stem cells had better results than the placebo group at both 9 weeks and 48 weeks. At 9 weeks, 59.5% of treated patients achieved that combined goal compared with 27.8% in the placebo group; at 48 weeks, 43.2% achieved it compared with 11.1%.
Good tolerability and favorable safety in cellular therapy for Type 2 diabetes
Having an alternative that can be evaluated not only for therapeutic intent but also for safety is especially important for people living with Type 2 diabetes who are seeking innovative options with clinical support. Xiao Fen Lian and team published a phase II clinical trial in World Journal of Clinical Cases (2023) to evaluate the safety of intravenous infusion of mesenchymal cells derived from human umbilical cord tissue in patients with Type 2 diabetes. During 24 weeks of follow-up, no serious adverse events were observed; in addition, there were no differences in tumor markers, no liver damage was detected, and no other side effects were seen on chest X-ray. Although 16.7% of patients experienced transient fever and one patient had hypoglycemia within the first month, the overall findings showed good tolerability of the treatment.
The cellular approach was administered with a clinically favorable safety profile, which is a key point when considering a high-specialty therapeutic alternative.
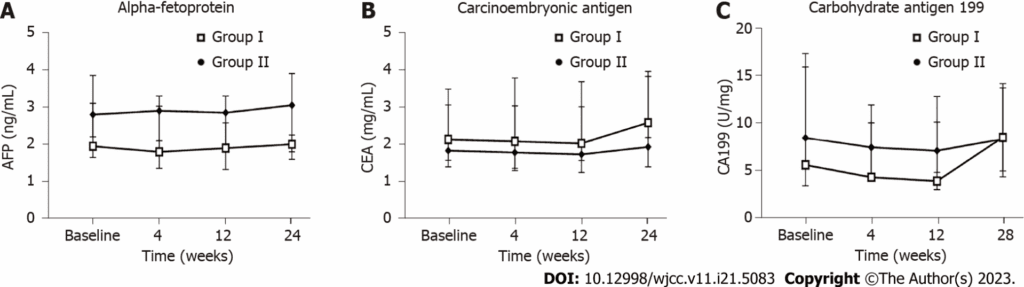
What this figure shows is that the tumor markers AFP, CEA, and CA199 remained without differences between the hUC-MSC-treated group and the placebo group at the evaluated follow-up points. This is important because, within a study focused on safety, the image helps support that no analytical signals suggesting a concerning trend appeared in this area during the observed period. The article itself complements this by stating that no nodules or tumors were detected in imaging studies performed during follow-up.
Discover why more and more people are turning to Biocelk® for a new possibility for their well-being.

