Why Biocelk?
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Explore Treatments
Scientific Literature
Featured Treatments
Wellness & Longevity
Dermo-aesthetic

Fertility
About Biocelk
Our Brands
Visit: regenivf.com
Osteoarticular and Orthopedic Injuries
This is a regenerative medicine protocol based on the application of mesenchymal stem cells, designed to improve the health of your joints, reduce pain, and help you recover your movement naturally.
Biocelk®’s osteoarticular treatment is designed for people living with joint pain or deterioration—whether due to wear and tear, inflammation, or injury—who want a non-invasive, science-backed alternative to improve their quality of life.
Clinical benefits
At Biocelk®, we take regenerative medicine further to help you recover movement, reduce pain, and give your body a more advanced opportunity to heal.
Knee Osteoarthritis
Living with knee osteoarthritis often means pain when walking, stiffness when getting up, and the feeling that every daily activity takes more effort.
Matas and collaborators published a phase I/II randomized controlled clinical trial in Stem Cells Translational Medicine (2019) that evaluated intra-articular injection of umbilical cord-derived mesenchymal stem cells, comparing a single dose versus repeated doses, and versus hyaluronic acid. Follow-up over 12 months showed no serious adverse events. Only the patients treated with stem cells showed significant improvements in pain and function; at 12 months, the repeated-dose group reported less pain than the comparison group.
Repeated application of stem cells may offer longer-lasting pain relief and improve mobility, without serious side effects. If you suffer from knee osteoarthritis, we invite you to explore with a specialist whether this treatment may be appropriate in your case.
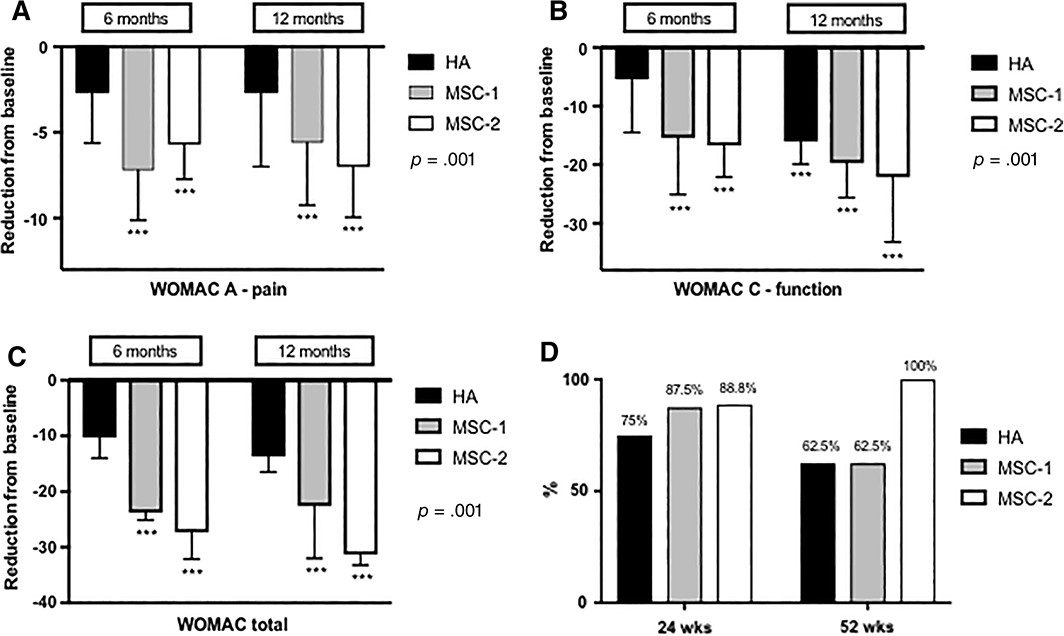
Clinical evolution of pain and function at 6 and 12 months
In this study, three treatments for knee osteoarthritis were compared: hyaluronic acid (HA), a single dose of umbilical cord-derived stem cells (MSC-1), and repeated doses of the same stem cells (MSC-2, administered at baseline and at 6 months). The results showed that the groups treated with stem cells (MSC-1 and MSC-2) improved in terms of pain and knee function, but those who received repeated doses (MSC-2) had the greatest improvement over time. At 12 months, those who received the repeated dose experienced less pain and greater improvement in mobility compared with the other groups. This suggests that repeated stem cell doses may be more effective for controlling pain and improving knee function over the long term.
When knee osteoarthritis progresses, pain while walking or climbing stairs and stiffness when standing up after sitting can become part of your routine, limiting simple daily activities.
Davatchi and collaborators published a long-term follow-up in the International Journal of Rheumatic Diseases on mesenchymal stem cell (MSC) therapy in knee osteoarthritis (2015). They reported a 5-year follow-up in three patients with moderate to severe osteoarthritis who received an intra-articular injection of 8–9 × 10⁶ mesenchymal stem cells in the most affected knee. At 6 months, the treated knees showed improvement in several clinical parameters (such as pain and function), and although there was gradual deterioration over time, at 5 years they still remained better than at baseline, in addition to improvement in the patient’s global assessment. The knee that did not receive cells continued to worsen and ultimately became the most affected. In practice, this suggests that the cellular approach may offer sustained clinical improvement, though not necessarily permanent, especially when applied in less advanced stages. If you live with limitations due to osteoarthritis, it is worth exploring with a specialist whether this kind of alternative could be evaluated in your case.
El estudio reporta una mejoría global a los 6 meses y, aunque algunos indicadores se deterioran gradualmente con el tiempo, a 5 años la rodilla tratada seguía mejor que al inicio, mientras que la rodilla contralateral sin MSC continuó progresando y terminó siendo la más afectada.
If you live with knee osteoarthritis, you know how heavy the pain of walking, the stiffness, and the feeling that every step takes a toll on your day can be.
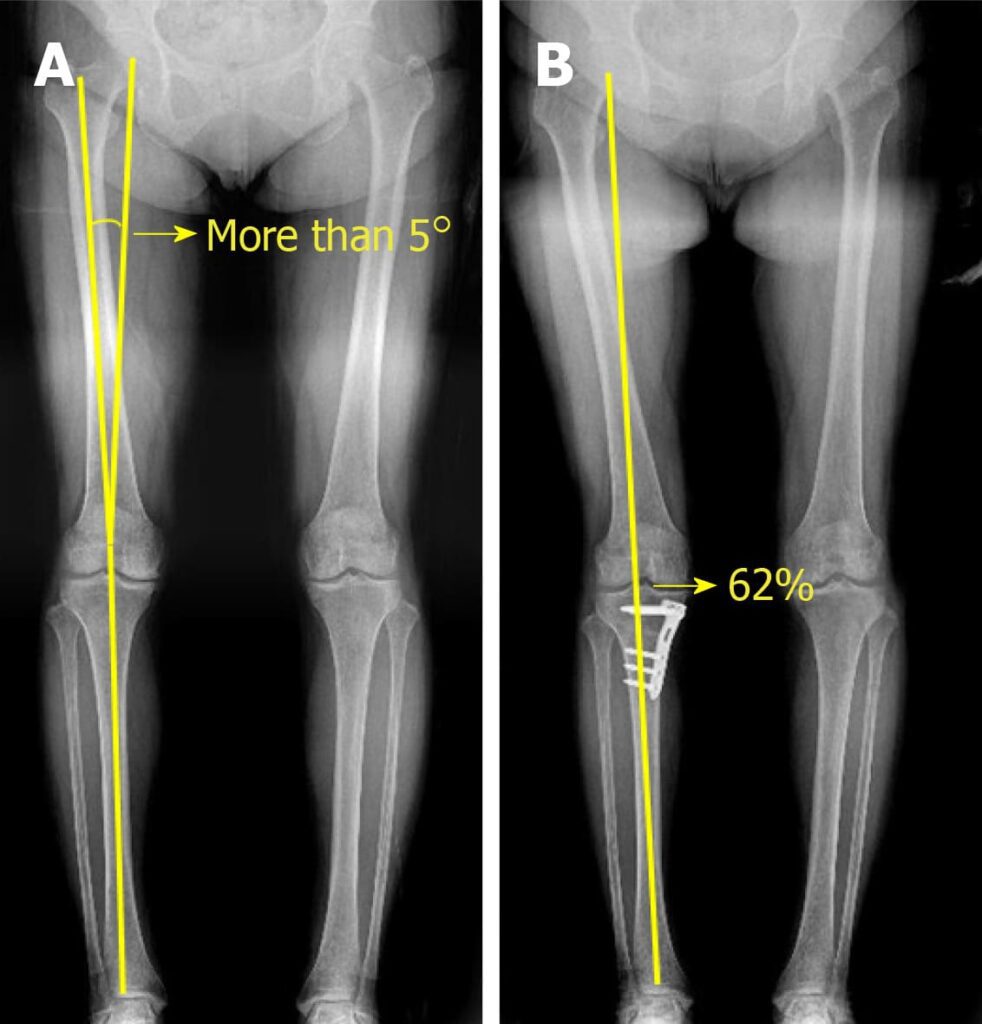
Arthroscopic comparison of knee cartilage before treatment and during follow-up after high tibial osteotomy combined with implantation of umbilical cord blood-derived mesenchymal stem cells. This indicates that the treatment helped the damaged cartilage areas begin to fill in with new tissue, which may contribute to improving joint function.
Xiao and collaborators published a systematic review with meta-analysis in Medicine (2024) evaluating the impact of UC-MSCs applied by intra-articular injection in knee osteoarthritis, integrating three randomized clinical trials (search up to March 31, 2024). Overall, the group treated with UC-MSCs showed a reduction in WOMAC versus control (approximately 25.85 points lower) and an improvement in Lysholm score (approximately 18.33 points higher). In practice, these findings suggest a reduction in pain and a perceived functional improvement after the intervention, although the evidence is still limited due to the low number of studies, and more research is needed to confirm it. If you are looking for options for your knee, it is worth exploring with a specialist whether this approach could be an alternative in your case.
The image shows that some patients moved from low scores to higher values on the erectile function questionnaire, which translates into better ability to achieve and maintain an erection during the study follow-up.
Discover how Biocelk® can help you regain freedom of movement with an approach designed around you.
Osteonecrosis
When advanced osteonecrosis of the femoral head causes pain and makes walking difficult, the greatest concern is usually the possibility of bone collapse and total hip replacement at a very young age.
Gómez-Barrena and collaborators published the ALOFEM pilot study in Stem Cells International (2026), in which they evaluated the feasibility, safety, and early efficacy of a treatment with allogeneic mesenchymal stem cells (MSCs) in young patients with severe corticosteroid-induced osteonecrosis. The treatment consisted of the surgical implantation of expanded MSCs in 7 hips from 4 patients, and the results showed that the procedure was safe and feasible at 1- and 4-year follow-up, with no serious adverse events. In addition, 5 of the 7 treated hips avoided total hip replacement at 4 years.
This suggests that this cellular approach could be an option to preserve the joint and delay major surgery. If osteonecrosis is part of your life, it is advisable to consult a specialist to see whether this treatment may be appropriate for your case.
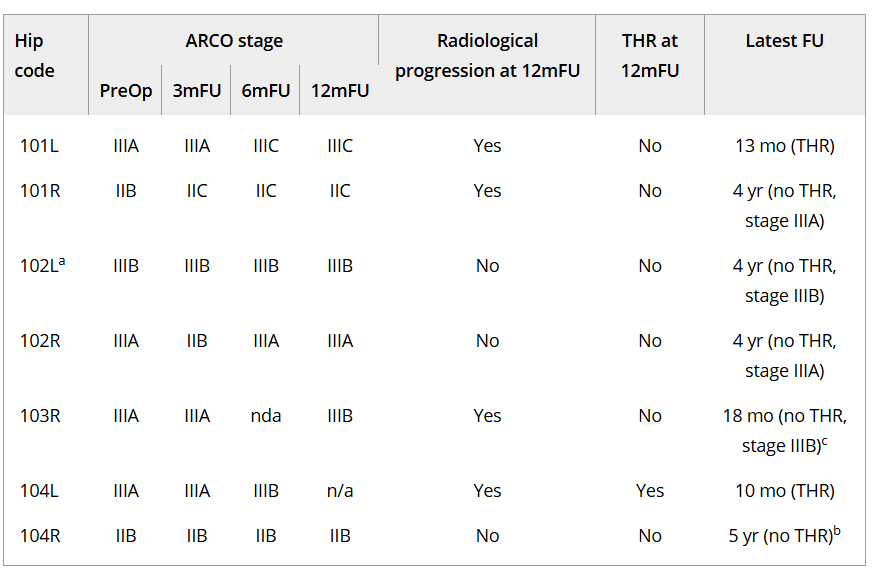
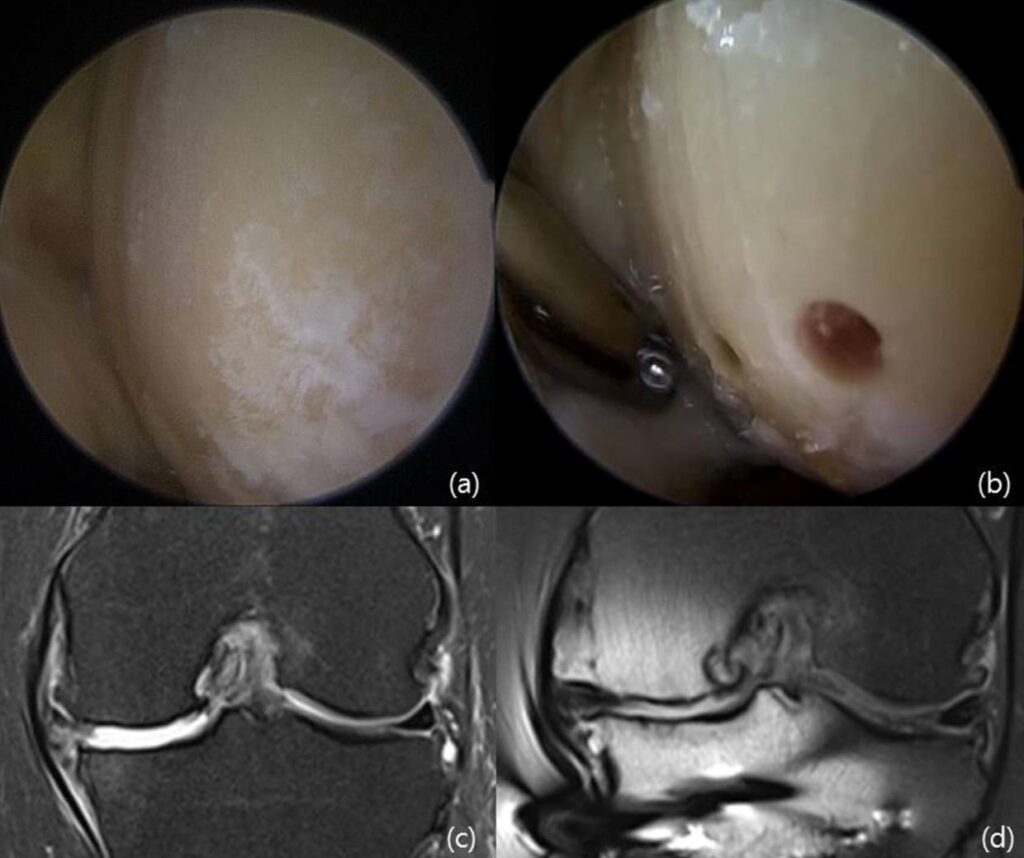
Seven treated hips were followed after local implantation of 140 × 10⁶ allogeneic MSCs. The table summarizes how ARCO stage evolved at the 3-, 6-, and 12-month controls, and whether there was conversion to total hip replacement (THR).
At 12 months, radiologic progression was reported in 4/7 hips, and 2 of them required THR (at 10 and 13 months). At the latest follow-up reported, 5/7 hips had avoided THR long term (up to 4–5 years, depending on the case), supporting the feasibility of the approach and its clinical potential to postpone major surgery in young patients with severe osteonecrosis.
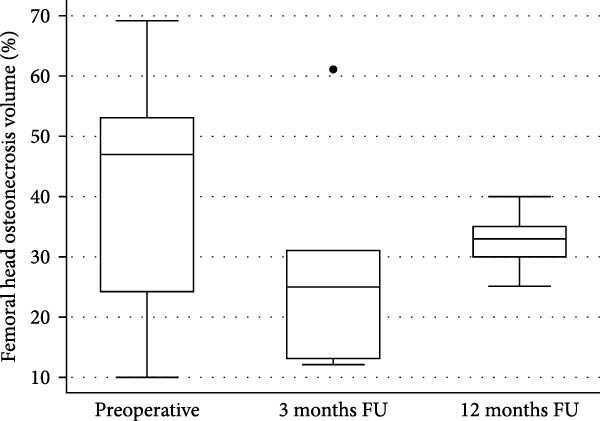
The figure shows ONV (osteonecrosis volume) as a percentage of the femoral head before the procedure and during follow-up. The reported median was 47% preoperatively, decreased to 25% at 3 months, and was 33% at 12 months.
In clinical terms, this image suggests an early reduction in osteonecrosis volume after cellular implantation, followed by a tendency to increase again toward one year. The study itself points out that, in this limited series, the observed reduction did not reach statistical significance versus the preoperative value at the evaluated time points.
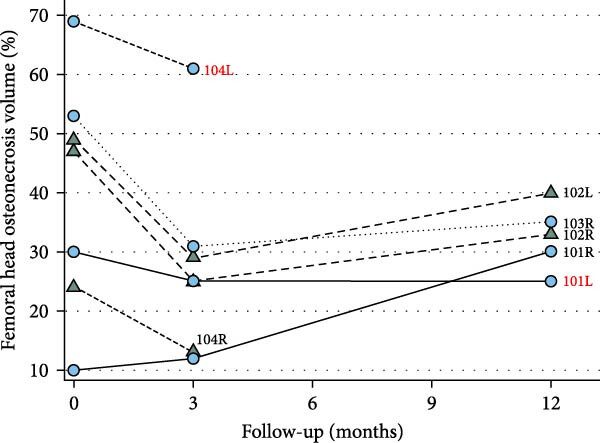
This graph allows hip-by-hip behavior to be visualized. In general, the study reports that almost all cases tended to show a decrease in ONV at 3 months, with one exception (a hip that did not show that initial reduction). By the 12-month control, the described trend is that ONV increases again.
The figure also visually differentiates hips with and without progression/THR (according to the symbols indicated in the article legend), offering a clear interpretation: the most consistent effect occurs early, while one-year follow-up shows a more heterogeneous scenario.
Schedule your evaluation and discover why Biocelk® is shaping a new way to support joint recovery.

