Why Biocelk?
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Personalized Diagnosis
Highly Qualified Medical Team and Personnel
Largest and Most Reliable Cell Bank in Latin America
Cutting-edge Technology
Explore Treatments
Scientific Literature
Featured Treatments
Wellness & Longevity
Dermo-aesthetic

Fertility
About Biocelk
Our Brands
Visit: regenivf.com
Obesidad y Metabolismo
At Biocelk®, metabolic medicine begins with a fundamental idea: health starts in the way your cells produce and use energy. When that balance is disrupted, the body begins to show it in different ways, from chronic inflammation, insulin resistance, and hormonal changes, to difficulty regulating weight, accelerated aging processes, or alterations in reproductive health. That is why this approach seeks to identify how your body is functioning at the cellular and metabolic level in order to intervene in a personalized, precise, and clinically grounded way.
Clinical Benfits
At Biocelk®, we treat metabolism from a deeper perspective to help you regain balance, improve the way your body responds, and build more stable health from within.
Hormonal Regulation
Improvements in sexual satisfaction
This suggests that this type of approach may support biological processes related to sexual response, helping restore part of the well-being and confidence involved in intimate life.
The treatment not only helped patients feel better, but also supported more favorable hormonal regulation, opening a more comprehensive possibility for restoring well-being.
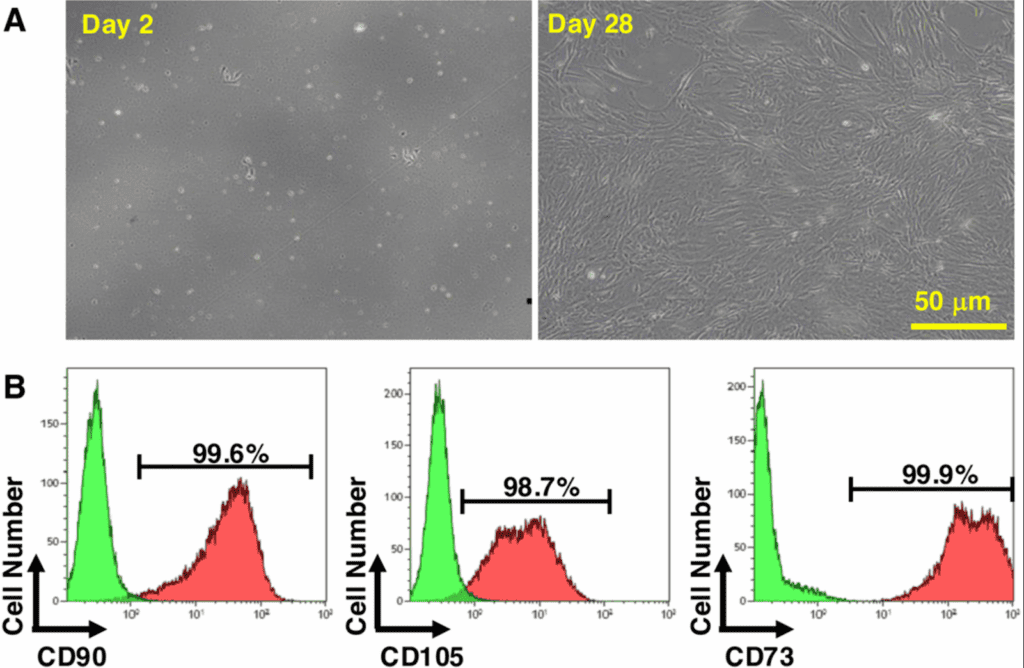
In panel (A), the microscopy images show how the cells evolve from day 2 to day 28 in culture. From early stages, they adopt a characteristic elongated, fibroblast-like morphology, and over time they form a more organized and homogeneous population. This pattern is not incidental: it is typical of mesenchymal stem cells under optimal conditions, indicating stability, viability, and cellular expansion capacity.
In panel (B), flow cytometry confirms the purity of the cellular population. The cells positively express markers such as CD73, CD90, and CD105, considered international standards for identifying mesenchymal stem cells. This validates that it is not an undefined cell mixture, but a well-characterized population.
If you are looking for much more than weight control, at Biocelk® you will find a different way to care for your body.
Glycemic Control
More patients achieved glycemic control and better blood stability with this cellular approach.
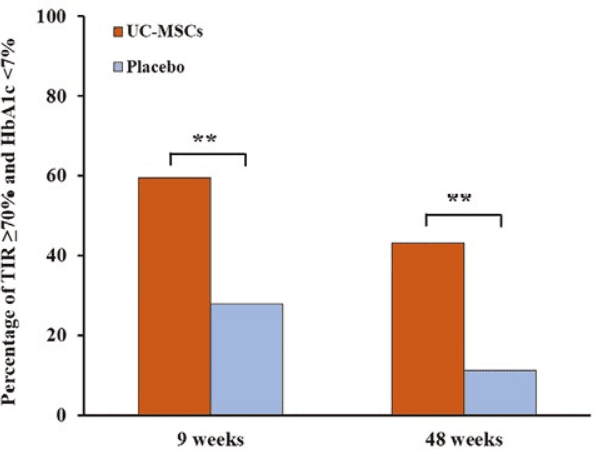
The figure shows that the group treated with mesenchymal stem cells derived from umbilical cord tissue had better results than the placebo group at both 9 weeks and 48 weeks. At 9 weeks, 59.5% of treated patients achieved that combined goal versus 27.8% with placebo; at 48 weeks, 43.2% achieved it versus 11.1%.
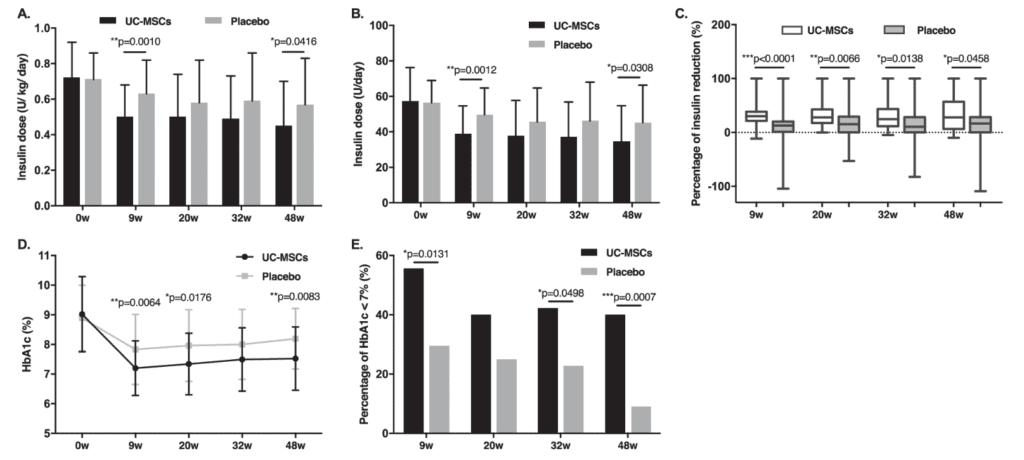
Living with type 2 diabetes is not just about dealing with high numbers: it also means managing glucose highs and lows that are exhausting, stressful, and increase the risk of complications over time. Zang and collaborators published a phase II randomized, placebo-controlled trial in Stem Cells Translational Medicine (2023), in which they evaluated intravenous infusion of mesenchymal stem cells derived from umbilical cord tissue in 73 adults with type 2 diabetes. After 48 weeks, the treated group showed more marked improvement than placebo in both time in glycemic range and HbA1c; in addition, more patients achieved the combined goal of TIR ≥70% and HbA1c <7% at weeks 9 and 48 (59.5% vs 27.8% and 43.2% vs 11.1%).
This suggests more stable blood glucose throughout the day, fewer harmful spikes, and more favorable metabolic control for living with greater peace of mind and long-term protection.
Insulin Resistance Modulation
- More patients were able to lower their glycated hemoglobin and cut their insulin use in half after treatment at Biocelk
Living with type 2 diabetes often means a constant routine of medications, adjustments, and the concern that, even with effort, metabolic control may still not be enough.
Li Zang and collaborators published a phase II randomized, double-blind, placebo-controlled trial in Stem Cell Research & Therapy (2022) in Chinese adults with type 2 diabetes, where they evaluated intravenous infusion of mesenchymal stem cells derived from umbilical cord tissue. At 48 weeks, 20% of treated patients achieved the combined goal of HbA1c below 7% and at least a 50% reduction in daily insulin, versus 4.55% in placebo; in addition, HbA1c decreased by 1.31% in the treated group, and insulin resistance also improved.
This suggests better blood sugar control with less dependence on insulin and a more favorable metabolic response, opening a real possibility of moving forward with greater stability.
If you are looking for regenerative alternatives for type 2 diabetes, at Biocelk we can help you evaluate your case.
Figure 2 focuses on two very clear outcomes: the decrease in insulin requirements and the reduction in HbA1c after infusion of UC-MSCs. The article describes that, compared with placebo, the treated group maintained lower insulin requirements at several follow-up points and showed a more marked reduction in HbA1c, with a decrease of 1.31% versus 0.63% in placebo at 48 weeks. In addition, 13.5% of treated patients became insulin-free, something that did not occur in the placebo group.
Reduction of Systemic Inflammation
Better quality of life, more strength, and better mobility in patients with aging-related frailty
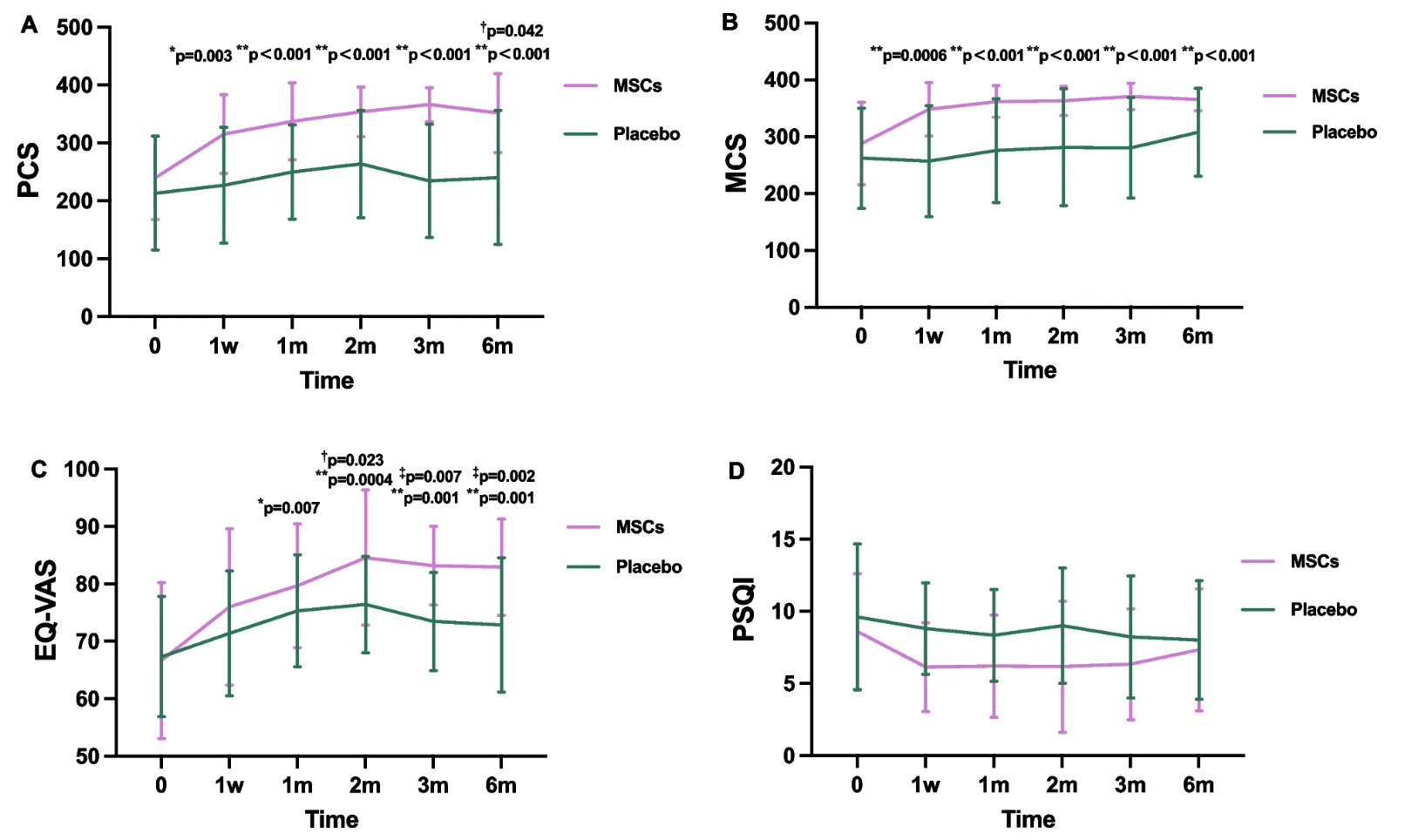
When the body begins to lose endurance, balance, and energy, it is not only movement that changes: confidence in daily life changes too. Zhu and collaborators published a phase I/II randomized, double-blind, placebo-controlled study in Stem Cell Research & Therapy (2024), in which they evaluated intravenous infusion of mesenchymal stem cells derived from umbilical cord tissue in patients with aging-related frailty. The results showed significant improvements in the physical component of quality of life from the first post-treatment visit and sustained through 6 months, as well as progress in functional tests such as Timed Up and Go and grip strength, especially toward the end of follow-up. TNF-α and IL-17 also decreased versus placebo, with no differences in adverse event incidence.
This suggests real support for regaining mobility, strength, and a sense of well-being, while reducing part of the chronic inflammation that accompanies physical decline.
The figure shows changes in quality of life: the group treated with HUC-MSCs improved significantly in the physical component of the SF-36 from the first post-treatment visit and maintained that improvement throughout follow-up; in addition, EQ-VAS was also better than placebo from month 2 through month 6. In simple terms, this figure suggests that patients were not only moving better, but also perceived themselves as being in better overall health.
Schedule your evaluation and discover how Biocelk® can help you transform your metabolic health from the root.

